UmCalahle ontuhlambili

umCalahle ontuhlambili ( ifomula yevanga CO2) ngumxube wevanga othalakala njengohowo oluyisimunyane olungenambala olunesiluthi esingaba amaphesenti angama-53 ngaphezulu kunalawo womoya owomile. AmaChembe womCalahle ontuhlambili aqukethe iChwe lomCalahle oluthungelwe ngokuphindiwe emaChweni amabili wesOhwe. UmCalahle ontuhlambili utholakala ngokwemvelo kumoyambulunga kaNomkhubulwane njenge njengohowo oluncane. Imithombo yawo engokwemvelo ihlanganisa izintaba-mlilo, ukusha kwamahlathi, iziphethu ezishisayo, ama-geyser, futhi uphuma nasematsheni asamCalahle ngokuncibilikiswa amanzi nayisimunyane. Ngenxa yokuthi umCalahle ontuhlambili uyancibilika emanzini, utholakala ngokwemvelo emanzini angaphansi komhlaba, imifula namachibi, izingqimba zeqhwa, izinguzunga zeqhwa nasemanzini olwandle. Uyatholakala nasezingxenyeni amafuthetshe nohowo lwemvelo. Umcalahle ontuhlambili unephunga elibukhali nelimunyu futhi ukhiqiza ukunambitheka kwamanzi ayiNaside emlonyeni. [1]Kodwa-ke, ekugxileni okuvamile awunaphunga.
Umlando[hlela | Hlela umthombo]

umCalahle ontuhlambili kwakunguhowo lokuqala oluchazwa njengesiqa esihlukile. Cishe ngo-1640, [2] umthaki wemithi Jan Baptist van Helmont waphawula ukuthi lapho eshisa isalahle esitsheni esivaliwe, inqumbi yomlotha yayiyiningi kunaleyo yesalahle yasekuqaleni. Isikhumusho sakhe sasiwukuthi enye ingxenye yesalahle idluliselwe esiqeni esingabonakali ayesibiza ngokuthi "uhowo" noma "umoya wasendle" (i- spiritus sylvestris ).
Izakhi zevanga nezokubumbeka[hlela | Hlela umthombo]
uhlaka, ukubhonqeka nokuqhikizeka kwamachembe[hlela | Hlela umthombo]
Umlinganiso wamachembe omcalahle ontuhlambili uluqombotha futhi ulingenindeni yomlingazwe wawo wokulingana. Ubude besibhobqi somcalahle-sOhwe kumcalahle ontuhlambili ungu-116.3 pm, mfushane ngokuphawulekayo kunobude obucishe bube ngu-140-pm besibhonqi esijwayelekile se-C-O, futhi sifushane kunamanye amaqembu amaningi asebenza ngokubhonqa ama-C-O njenge- carbonyls. Njengoba lilingenindeni, ichembe alinaso isikhashana se-dipole yamazuba.

Kwisixazululo samanzi[hlela | Hlela umthombo]
umCalahle ontuhlambili uyancibilika emanzini, lapho ubuyela emuva ubuye ubumbe H</br> H CO</br> H (isimunyane somcalahle), okuyisimunyane esibuthakathaka ngoba ukuhonyozeka kwaso emanzini kunga.
- CO2 + H2O ⇌ H2CO3
Ukuhlala kwe-hydration equilibrium kwe-carbonic acid kuyinto (ngo-25 ° C). Ngakho-ke, iningi lomcalahle ontuhlambili aliguqulwa libe yi-carbonic acid, kepha lihlala njengama- molecule e-CO 2, angathinti i-pH.
Ukugxila okuhlobene kwe- CO</br> CO, HCO CO CO kanye deprotonated amafomu HCO-</br> HCO ( bicarbonate ) ne- CO2−</br> CO ( carbonate ) incike kwi- pH.Njengoba kukhonjisiwe esakhiweni seBjerrum, emanzini angathathi hlangothi noma ane-alkaline encane (pH> 6.5), ifomu le-bicarbonate ligcwele (> 50%) liba kakhulu (> 95%) ku-pH yamanzi olwandle.Emanzini ane-alkaline kakhulu (pH> 10.4), ifomu elivelele (> 50%) yi-carbonate.Izilwandle, njengoba zine-alkaline emnene ene-pH ejwayelekile = 8.2-8.5, ziqukethe cishe i-120 mg we-bicarbonate ngelitha ngalinye.
Ukuba yi- diprotic, i-carbonic acid inezakhi ezimbili zokuhlukaniswa kwe-asidi, eyokuqala yokuhlukaniswa kwe-bicarbonate (ebizwa nangokuthi i-hydrogen carbonate) ion (HCO 3 - ):
- H 2 CO 3 ⇌ HCO 3 - + H +
- K a1 = 2.5× 10 −4 mol / L ; p K a1 = 3.6 ku-25 ° C.
Lokhu kungukuhlala kweqiniso kwe-acid dissociation okuchazwa njenge , lapho i-denominator ifaka kuphela i-covalently binded H 2 CO 3 futhi ingafaki i-hydrated CO 2 (aq). Inani elincanyana kakhulu futhi elicashunwa njalo eliseduze no- 4.16× 10 −7 liyinani elibonakalayo elibalwe ekucabangeni (okungalungile) kokuthi yonke i-CO 2 encibilikile ikhona njenge-carbonic acid, ukuze . Njengoba iningi le-CO 2 elichithakele lihlala njengamamolekyu e-CO 2, i- K a1 (ebonakalayo) inesilinganiso esikhulu kakhulu nenani elincane kakhulu kune- K a1 yangempela.
I- bicarbonate ion iyinhlobo ye- amphoteric engasebenza njenge-asidi noma njengesisekelo, kuya nge-pH yesisombululo.Ku- pH ephezulu, yehlukanisa kakhulu ne- carbonate ion (CO 3 2− ):
- I-HCO 3 - ⇌ CO 3 2− + H +
- K a2 = 4.69× 10 -11 mol / L ; p K a2 = 10.329
Ezidalweni ukukhiqizwa kwe-carbonic acid kukhishwa yi- enzyme, i- carbonic anhydrase.
Izakhiwo zomzimba[hlela | Hlela umthombo]

I-Carbon dioxide ayinambala. Ezingxenyeni eziphansi igesi ayinaphunga; noma kunjalo, ekugxileni okwanele ngokwanele, inephunga elibukhali, eline-esidi.Ekushiseni okujwayelekile nokucindezela, ubukhulu be-carbon dioxide buzungeze u-1.98 kg / m 3, cishe izikhathi eziyi-1.53 zomoya.
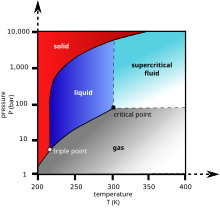
ICarbon dioxide ayinaso isimo 0.517engaphansi kuka-0.517 95 (10) I-MPa [3] ( 5.11177 (99) i-atm ). 0.101 kwe-1 atm ( 0.101 MPa ), igesi ifaka ngqo esiqinile emazingeni okushisa angaphansi kuka- 194.6855(30) I-K [4] ( −78.4645(30) ° C ) kanye nezimbumbulu eziqinile ngqo kwigesi engaphezulu kwaleli zinga lokushisa. Esimeni sayo esiqinile, isikhutha ngokuvamile ibizwa ngokuthi yiqhwa elomile .
Izicelo[hlela | Hlela umthombo]
ICarbon dioxide isetshenziswa yimboni yokudla, imboni kawoyela, kanye nemboni yamakhemikhali. [5]Le nhlanganisela inokusetshenziswa okuhlukahlukene kokuhweba kepha okunye ukusetshenziswa kwayo okukhulu njengekhemikhali ukukhiqizwa kweziphuzo ezinekhabhoni; inikeza ukukhanya kweziphuzo ezine-carbonated ezifana namanzi we-soda, ubhiya newayini elikhazimulayo.
Isandulela samakhemikhali[hlela | Hlela umthombo]
Embonini yamakhemikhali, isikhutha sidliwa kakhulu njengesithako ekukhiqizeni i- urea, kusetshenziswa ingxenyana encane ukukhiqiza i- methanol nohla lweminye imikhiqizo.Ezinye izakhi ze-carboxylic acid ezifana ne- sodium salicylate zilungiswa kusetshenziswa i-CO 2 yi- Kolbe-Schmitt reaction.
Ngaphezu kwezinqubo ezijwayelekile ezisebenzisa i-CO 2 yokukhiqiza amakhemikhali, izindlela ze-electrochemical nazo ziyahlolwa ezingeni locwaningo.Ikakhulu, ukusetshenziswa kwamandla avuselelekayo ekukhiqizeni uphethiloli ovela ku-CO 2 (njenge-methanol) kuyaheha ngoba lokhu kungaholela ekubaseni okungathuthwa kalula futhi kusetshenziswe ngaphakathi kobuchwepheshe obuvamile bomlilo kepha kungabikho ukukhishwa kwe- CO 2. [6]
Izitshalo zidinga i-carbon dioxide ukuqhuba i- photosynthesis.Umkhathi wezindawo zokugcina izithombo (uma ngabe usayizi omkhulu, kufanele) ucebiswe nge-CO 2 eyengeziwe ukugcina nokukhuphula izinga lokukhula kwezitshalo.Ngezinye lokuhlala eliphezulu kakhulu (izikhathi 100 lokuhlushwa asemkhathini, noma ngaphezulu), i-carbon dioxide angaba enobuthi ekuphileni isilwane, ngakho nokuvusa okuhlushwa ukuze 10,000 ppm (1%) noma ngaphezulu amahora ambalwa uyoqeda izinambuzane ezifana whiteflies futhi izibungu spider endaweni abamba ukushisa. [7]
Ekudleni[hlela | Hlela umthombo]

Izinkomba[hlela | Hlela umthombo]
- ↑ Carbonated (Sparkling) Water: Good or Bad? healthline.com
- ↑ DavidFraser Harris (September 1910). The Pioneer in the Hygiene of Ventilation. https://zenodo.org/record/2088803.
- ↑ Span, Roland; Wagner, Wolfgang (1996-11-01). "A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple‐Point Temperature to 1100 K at Pressures up to 800 MPa". Journal of Physical and Chemical Reference Data 25 (6): 1519. Bibcode 1996JPCRD..25.1509S. doi:10.1063/1.555991. ISSN 0047-2689. Archived from the original on 2021-07-09. https://web.archive.org/web/20210709182537/https://aip.scitation.org/doi/10.1063/1.555991. Retrieved 2021-09-10.
- ↑ Span, Roland; Wagner, Wolfgang (1996-11-01). "A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple‐Point Temperature to 1100 K at Pressures up to 800 MPa". Journal of Physical and Chemical Reference Data 25 (6): 1521. Bibcode 1996JPCRD..25.1509S. doi:10.1063/1.555991. ISSN 0047-2689. Archived from the original on 2021-07-09. https://web.archive.org/web/20210709182537/https://aip.scitation.org/doi/10.1063/1.555991. Retrieved 2021-09-10.
- ↑ Pierantozzi, Ronald (2001). Kirk-Othmer Encyclopedia of Chemical Technology. Wiley. doi:10.1002/0471238961.0301180216090518.a01.pub2. ISBN 978-0-471-23896-6.
- ↑ Badwal, Sukhvinder P. S.; Giddey, Sarbjit S.; Munnings, Christopher; Bhatt, Anand I.; Hollenkamp, Anthony F. (24 September 2014). "Emerging electrochemical energy conversion and storage technologies (open access)". Frontiers in Chemistry 2: 79. Bibcode 2014FrCh....2...79B. doi:10.3389/fchem.2014.00079. PMC 4174133. PMID 25309898. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=4174133.
- ↑ Stafford, Ned (7 February 2007). "Future crops: The other greenhouse effect". Nature 448 (7153). Bibcode 2007Natur.448..526S. doi:10.1038/448526a.

![{\displaystyle K_{\mathrm {h} }={\frac {\rm {[H_{2}CO_{3}]}}{\rm {[CO_{2}(aq)]}}}=1.70\times 10^{-3}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/880be9280cefa51b14dc80f037e4296c81a15e6b)
![{\displaystyle K_{a1}={\frac {\rm {[HCO_{3}^{-}][H^{+}]}}{\rm {[H_{2}CO_{3}]}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/81950791f8b36374241d6fa82828522bd875668a)
![{\displaystyle K_{\mathrm {a1} }{\rm {(apparent)}}={\frac {\rm {[HCO_{3}^{-}][H^{+}]}}{\rm {[H_{2}CO_{3}]+[CO_{2}(aq)]}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d4cac9482fd21cb58444f2621f245d7b1d835485)